- This event has passed.
Prof. Kana M. Sureshan “Synthesis of Biopolymer Mimics via Topochemical Reactions”
June 11, 2019 @ 11:00 am
Kana M. Sureshan
Indian Institute of Science Education and Research Thiruvananthapuram, India
kms@iisertvm.ac.in ; sureshankana@gmail.com
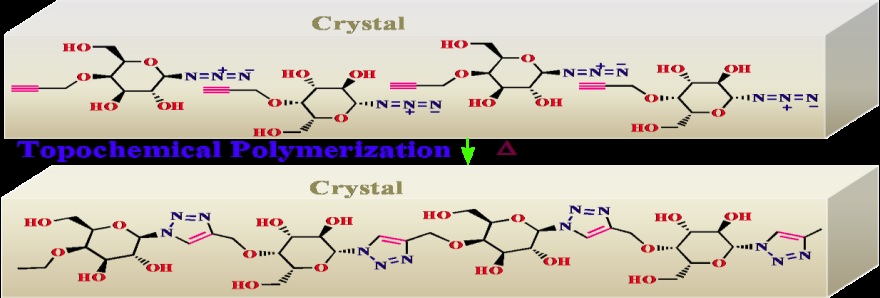
Topochemical reactions, the reaction between pre-organized reacting motifs in the crystal lattice, are attractive as they do not require solvents, catalysts and other special reaction conditions for the reaction and provide products in pure form. Also such reactions provide basic understanding about mechanistic and geometrical details about a reaction.1 We have employed thermal Topochemical Azide–Alkyne Cycloaddition (TAAC) reaction and light induced topochemical polymerization of diacetylenes to polydiacetylenes (PDA) to synthesize various biopolymer mimics. We have exploited hydrogen bonding for the self-assembly of monomers in solid or gel to pre-organize the reacting motifs. Lattice controlled polymerization/oligomerization reaction of such self-assembled (pre-organized) monomers gave various biopolymer mimics. By applying this methodology, we have topochemically synthesized monodisperse glycopolymers,2,3 oligosaccharide mimics,4 DNA analogs5,6 and polypeptides.7-12 Also we have exploited light-induced topochemical polymerization of diynes to polydiacetylenes for various applications.13-14 In this talk, the design and execution of such topochemical syntheses and their possible applications will be discussed.
References:
1 Photochemistry in Organized and Constrained Media, ed. Ramamurthy, V. VCH Publishers, New York, 1991
2 A. Pathigoolla, R. G. Gonnade, K. M. Sureshan, Angew. Chem. Int. Ed., 2012, 51, 4362.
3 A. Pathigoolla, K. M. Sureshan, Angew. Chem. Int. Ed., 2013, 52 8671.
4 K. Hema, K. M. Sureshan, CrystEngComm 2018, 20, 1478.
5 A. Pathigoolla, K. M. Sureshan, Angew. Chem. Int. Ed., 2014, 53, 9522.
6 A. Pathigoolla, K. M. Sureshan, Chem. Commun. 2016, 52, 886.
7 B. P. Krishnan, R. Rai, A. Asokan, K. M. Sureshan, J. Am. Chem. Soc., 2016, 138, 14824.
8 B. P Krishnan, K. M. Sureshan J. Am. Chem. Soc., 2017, 139, 1584.
9 R. Rai, B. P. Krishnan, K. M. Sureshan, Proc. Natl. Acad. Sci. U. S. A. 2018, 115, 2896.
10 R. Mohanarao, K. M. Sureshan, Angew. Chem. Int. Ed., 2018, 57, 12435.
11 V. Athiyarath, K. M. Sureshan, Angew. Chem. Int. Ed., 2019, 58, 612.
12 K. Hema, K. M. Sureshan, Angew. Chem. Int. Ed., 2019, 58, 2754.
13 B. P. Krishnan, S. Mukherjee, P. M. Aneesh, M. A. G. Namboothiry, K. M. Sureshan, Angew. Chem. Int. Ed. 2016, 55, 2345.
14 B. P Krishnan, S. Raghu, S. Mukherjee, K. M Sureshan, Chem. Commun., 2016, 52, 14089.